How a Cancer Drug Gets to Market
By Catherine Williams, Illustration by Damien Scogin
Conducting trials of new drugs and treatments goes to the heart and soul of Winship's mission to turn scientific discoveries into therapies that help cancer patients. The trials are painstaking. It can take nine or more years from the first time a new anticancer compound is tested in a lab, to the point when the U.S. Food and Drug Administration (FDA) grants permission to market it as a drug.
The 21st Century Cures Act signed at the end of 2016 proposes to speed up the FDA's drug approval process. But can that happen without sacrificing safety? How do we, the lay public, evaluate the proposed changes?
R. Donald Harvey, Winship's top pharmacy expert and director of Winship's Phase I Clinical Trials section, has a mixed response to the proposal: "There are no shortcuts to testing safety and efficacy, but the process could be better tailored to produce more types of information in less time."
 |
|---|
YEARS 1 — 2
Preclinical testing
Long before a new drug gets to the multi-phase clinical trial process, it is exhaustively tested in the lab using cell lines and animals. At this point, the "drug" is a collection of five to ten potential molecules that researchers have identified for their ability to impair or kill cancer. Testing involves administering escalating doses to animals in order to determine toxicity over the short term and long term. Researchers look at how the drug is metabolized, how it affects vital organs like bone marrow, liver, kidneys, and heart, and whether it causes mutations or interferes with fetal development.
"The biggest thing to learn from preclinical testing is what compounds to not pursue. If a drug is too toxic or doesn't work in an animal, then it's clearly something we shouldn't take into human testing." -R. Donald Harvey
Preclinical testing gathers the data that's crucial for the next step: a Phase I trial, the first-in-human testing.
 |
|---|
YEARS 3 — 4
Phase I clinical trial
To launch the clinical trial process, the drug's sponsor makes an investigational new drug (IND) application to the FDA. The IND contains all of the chemistry and preclinical testing data plus a protocol for how the Phase I clinical trial will be conducted. Usually, the IND is submitted by a pharmaceutical company that hopes to bring the drug to market. Once the IND is approved, clinical testing begins.
"Phase I trials that introduce a promising first-in-class molecule can be very exciting for Winship patients. These are the kind of trials we want to be involved in." -R. Donald Harvey
The primary goals of Phase I trials are to determine a safe dosage range and to see what the drug does in patients. How is it handled? Does it get to where it needs to be in the body? Does it cause side effects? Does it affect the cancer? Phase I trials typically enroll 15 to 30 patients who are divided into cohorts that receive the drug in different doses. The idea is to find the most effective dose that doesn't cause severe side effects.
 |
|---|
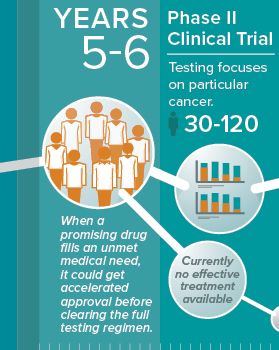
YEARS 5 — 6
Phase II clinical trial
Phase II trials hone in on how well the drug works on a particular cancer. They enroll 30 to 120 patients with the same type of cancer and set out to measure the success of the drug with that population. If there is currently no effective treatment for that cancer, then the drug might be eligible for accelerated approval (see graphic).
"One of the challenges with accelerating the process is you still have to compare the new treatment to what is already being done." -R. Donald Harvey
 |
|---|
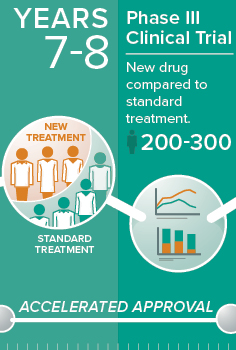
YEARS 7 — 8
Phase III clinical trial
This is where the rubber hits the road, when the new drug is compared to standard treatment already used in clinics.
"Sometimes the promising results from a Phase II trial don't hold up in a Phase III trial."& -R. Donald Harvey
Phase III trials enroll several hundred patients who are randomized to receive either the new drug or a standard treatment. The results of a Phase III trial should show how effective the new drug is and whether it's a viable alternative to what's already available.
 |
|---|
YEAR 9 AND BEYOND
New drug application
Once all testing data have been gathered and analyzed, the drug sponsor submits a new drug application (NDA) to the FDA. A team of scientists within the FDA reviews the testing data, the proposed drug labeling, and how the drug is to be manufactured, processed, and packaged. After a drug is approved, the FDA relies on a voluntary system of physicians and patients reporting adverse effects. Harvey would like to see more of this information gathered earlier, during the approval process.
"True personalization of treatment is understanding the right dose, given for the right duration, in the right patient." -R. Donald Harvey
Harvey says many factors go into how well a treatment works, such as how the drug interacts with foods and over-the-counter drugs and whether a patient's age or sex affects how it's metabolized. The goal is to find the best treatment for each individual patient, Harvey says, "We still have work to do to achieve this goal."